Compression Stroke
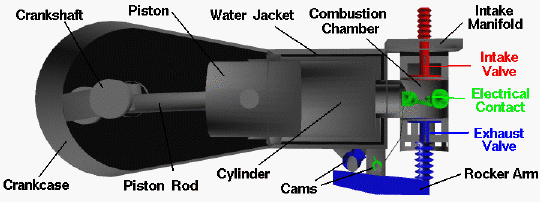
This is a computer drawing of one cylinder of the Wright brothers’ 1903 aircraft engine. This engine powered the first, heavier than air, self-propelled, maneuverable, piloted aircraft; the Wright 1903 Flyer. The engine consisted of four cylinders like the one shown above, with each piston connected to a common crankshaft which turns the propellers to produce thrust.
The brothers’ design is very simple by today’s standards, so it is a good engine for students to study to learn the fundamentals of engine operation. This type of internal combustion engine is called a four-stroke engine because there are four movements (strokes) of the piston before the entire engine firing sequence is repeated. In the figure, we have colored the fuel/air intake system red, the electrical system green, and the exhaust system blue. We also represent the fuel/air mixture and the exhaust gases by small colored balls to show how these gases move through the engine. Since we will be referring to the movement of various engine parts, here is a figure showing the names of the parts:
Mechanical Operation
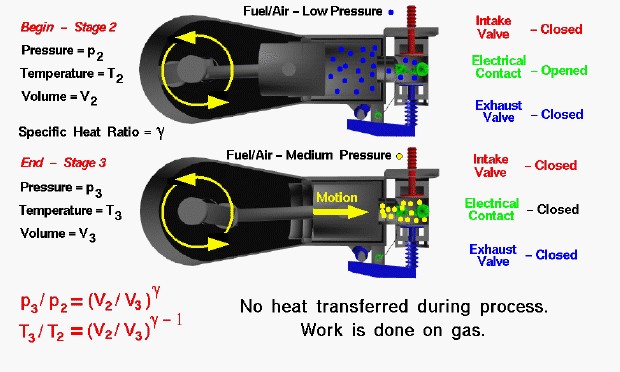
At the end of the intake stroke the fuel/air mixture has been drawn into the cylinder at low (nearly atmospheric) pressure by the motion of the piston towards the crankshaft at the left. From our considerations of the engine cycle, we designate this condition as Stage 2 of the Otto cycle. The intake valve is then closed and the piston begins to move back towards the combustion chamber at the right. With both valves closed, the combination of the cylinder and combustion chamber form a completely closed vessel containing the fuel/air mixture. As the piston is pushed to the right, the volume is reduced and the fuel/air mixture is compressed. When the piston has moved completely to the right, we designate the conditions as Stage 3 of the cycle. During the compression stroke, the electrical contact is kept opened. When the volume is the smallest, and the pressure the highest, the contact is closed and current flows through the completed circuit. The switch is then quickly opened, producing a spark which ignites the mixture.
Thermodynamics
During the compression, no heat is transferred to the fuel/air mixture. As the volume is decreased because of the piston’s motion, the pressure in the gas is increased. In the figure, the mixture has been colored blue at stage 2 and yellow at stage 3 to denote a moderate increase in pressure. To produce the increased pressure, we have to do work on the mixture, just as you have to do work to inflate a bicycle tire using a pump. There are thermodynamic equations which relate the pressure increase and temperature increase to the change in volume:
\(\LARGE \frac{p_3}{p_2}=\left(\frac{V_2}{V_3}\right)^\gamma\)
\(\LARGE \frac{T_3}{T_2}=\left(\frac{V_2}{V_3}\right)^{\gamma-1}\)
where p is the pressure, T is the temperature, V is the volume of the mixture, and gamma is the ratio of specific heats of the mixture. The numbers indicate the two stages of the cycle. Since V2 is greater than V3 and gamma (γ) is greater than 1 (1.4 for pure air), then p3 is greater than p2 and T3 is greater than T2. Pressure and temperature of the fuel/air mixture both increase during the compression process, and the final value (p3 and T3) depends only on a geometric compression ratio (V2/V3) to some power multiplied by the initial value (p2 and T2).